Clinical Applications
Heart Diseases
Heart muscle degeneration is the leading cause of debilitation and death in humans. It is the common pathway underlying congenital and infectious cardiomyopathies, myocardial infarction, congestive heart failure, angina, coronary artery disease and peripheral vascular disease, all of which constitute cardiovascular diseases. Global healthcare spending on cardiovascular diseases topped US$280 billion in 2001. In the US alone, approximately US$186 billion is spent every year in treating some 60 million cardiovascular disease patients. About 50% of the patients suffering congestive heart failure will die within five years of diagnosis.
Natural Heart Muscle Repair
Heart muscle degeneration results in loss of live cardiomyocytes, contractile filaments, contractility, heart function and healthy circulation. The damaged heart responds by cell division of cardiomyocytes. However, such regenerative capacity is hardly significant. Cardiomyocytes in culturo will undergo no more than three to five divisions, yielding an insufficient number of cells to repopulate any myocardial infarct.
Cardiomyocytes do not multiply significantly because the human telomeric DNA repeats in these terminally differentiated cells are minimal. Telomerasing cardiomyocytes in vivo still remains a technical challenge. Without significant mitotic activity, surviving cardiomyocytes cannot provide enough new cells to deposit the contractile filaments necessary to maintain normal heart function.
The degenerative heart also transmits biochemical signals to recruit stem cells from the stroma and the bone marrow in an attempt to repair the muscle damage. Much of the recruited stem cells differentiate to become fibroblasts instead of cardiomyocytes, thus forming fibrous scars and not contractile filaments. Thus, despite the claimed success of transmyocardial revascularisation using laser, angiogenic factors and genes, the damaged myocardium needs additional live myogenic cells to deposit contractile filaments to regain heart function, preferably before fibroblast infiltration, which leads to scar formation.
The Regenerative Heart
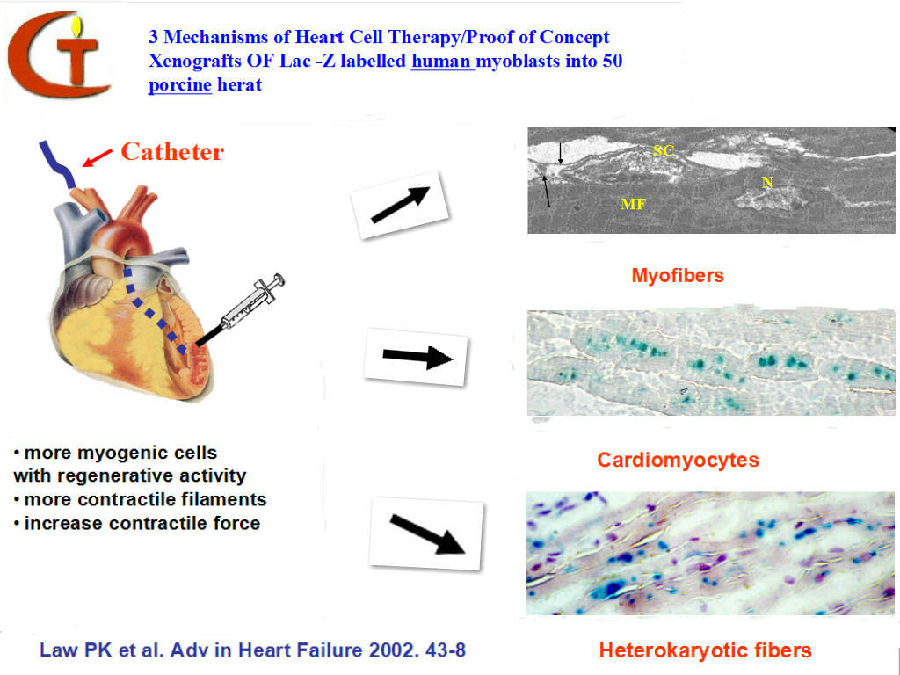
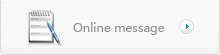
The goal to bioengineer the regenerative heart seems to be within reach with MTT. Five grams of muscle biopsies would be taken from both quadriceps of a heart patient with age ranging from 40 to 90, culturing approximately one billion myoblasts in four weeks and then injecting or surgically implanting these cells between the vascularised and the nonvascularised infracted myocardium.
Heart cell therapy (HCT), as this is called, is administered with the vision that the myoblasts will survive, develop and function as 'aliens' in the heart and their nuclei as aliens within cardiomyocytes. The myocardial aliens are newly formed skeletal myofibres that contribute to cardiac output through production of contractile filaments. They are donor in origin and, as skeletal myofibres, will have satellite cells and regenerative capability. The cardiomyocyte aliens are donor myoblast nuclei carrying chromosomes with long telomeric DNA subunits that are essential for mitosis. Upon injury of this heterokaryotic cardiomyocyte, the myoblast regenerative genome will be activated to produce foreign contractile filaments such as myosin.